
DBT-SAHAJ National Facility for
Single-Molecule and Super-Resolution Imaging


Technologies available
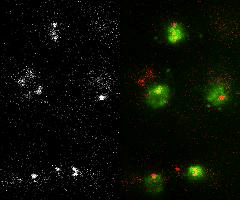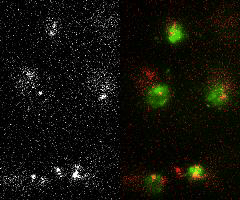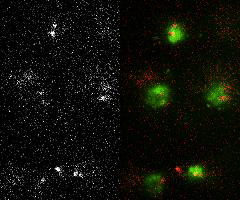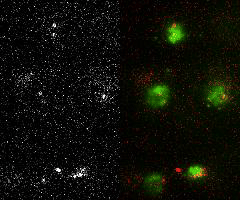
Single-molecule imaging and tracking (SMIT) in live cells to quantify protein dynamics and target-search mechanisms in live cells
Single-molecule imaging and tracking (SMIT) is a powerful tool for examining the biological structure and function of individual molecules. It offers a level of detail not achievable in ensemble studies, as it uncovers molecular behaviors that are otherwise hidden by the averaging of data. It further proposes—
• Diffusion dynamics of proteins in live cells
• Target-search mechanism of proteins
• Residence time of protein binding
• Fraction of bound and unbound molecules
Image Courtesy: Mehta's Lab
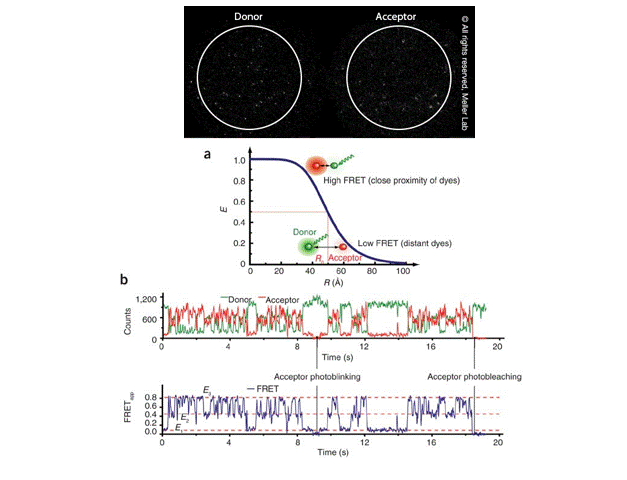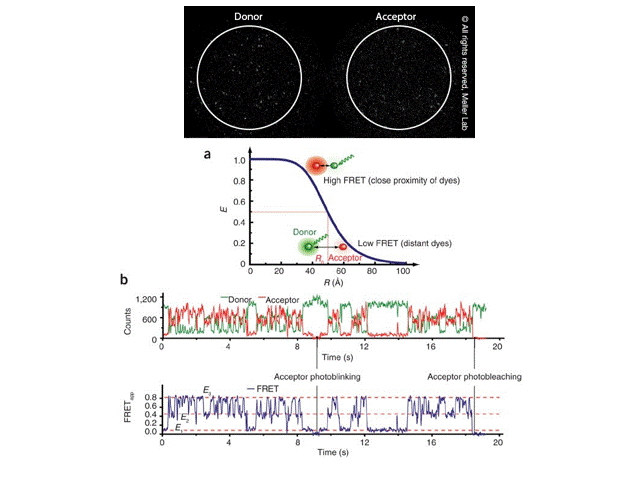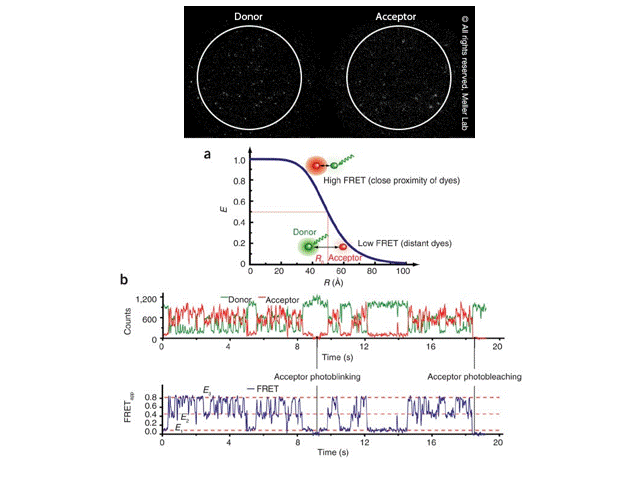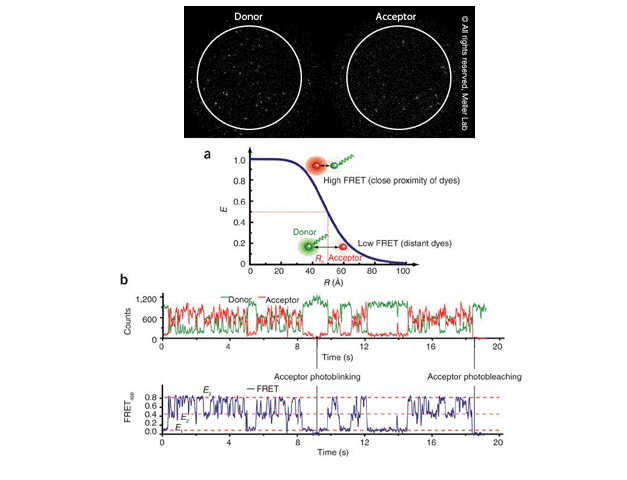
Single-molecule FRET (smFRET) for studying protein conformational dynamics
Single-molecule FRET (smFRET) has become a valuable technique for measuring changes in the distance between two molecules within the 10-100 Å range, which is highly relevant to molecular biological processes. It offers —
-
Protein conformation dynamics
-
Biomolecular interaction dynamics
Image Courtesy: Meller Lab and Roy et al 2018
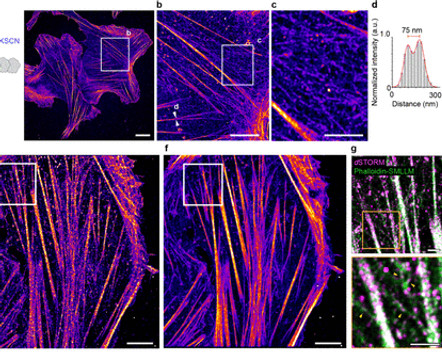
Single-molecule Localization Microscopy (SMLM), STORM, PALM, for high-resolution structure imaging (x,y resolution: 20 nm; z resolution: 50 nm)
Single-molecule localization microscopy (SMLM) encompasses a family of advanced imaging techniques that significantly enhance spatial resolution compared to standard diffraction-limited microscopy, allowing for visualization of biological structures at the molecular level..
Stochastic Optical Reconstruction Microscopy (STORM) creates a super-resolution image by accurately localizing individual fluorophores in three dimensions and across multiple colors It also can generate images of molecular structures resolved to under 20 nm.
In PALM microscopy, photoactivatable fluorophores are activated by lasers to resolve fine spatial details of tightly packed molecules. These fluorophores emit briefly and bleach, with the laser selectively activating random fluorophores in small groups to map their positions accurately.
All these advanced microscopic techniques offer—
-
High-resolution localization and structure analysis
-
Molecular clustering
-
Stoichiometry
Image Courtesy: Zongmin Zhao et al 2024

Single-molecule Fluorescence In Situ Hybridization (smFISH) to quantify transcription heterogeneity in the cell population
Single-molecule fluorescence in situ hybridization (smFISH) — also known as smRNA FISH or RNA FISH — is a cutting-edge technique for studying gene expression in single cells. It enables visualization and quantification of specifically labeled RNA molecules at single-molecule resolution; and offers—
-
Quantification of transcription dynamics and regulation
Image Courtesy: Evelina Tutucci et al 2019

6D imaging (x, y, z, time, multichannel, multiposition) in live and fixed cells.
6D super-resolution imaging (by combining 6 dimensions) provides a comprehensive view of complex biological systems, enabling researchers to study spatial structures, dynamic interactions, multi-channel (color) information, and molecular orientations. It proposes —
-
x, y, z, time, channels imaging altogether
-
Live cell imaging
-
Studying dynamics of cellular processes
Image Courtesy: Salgado-Almario J et al 2022
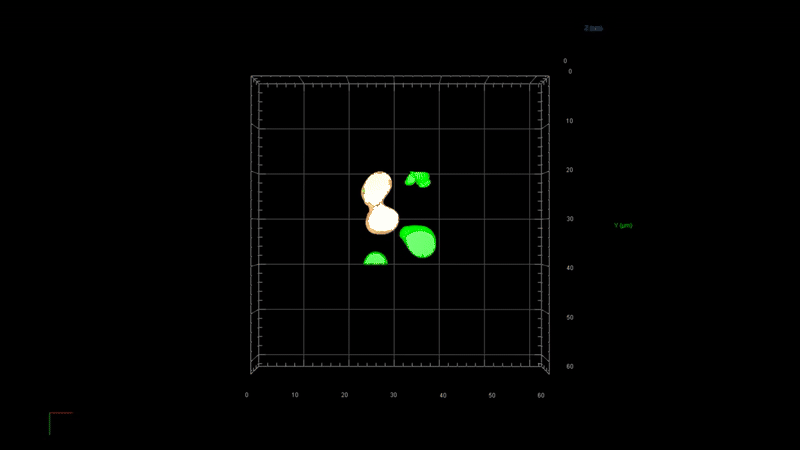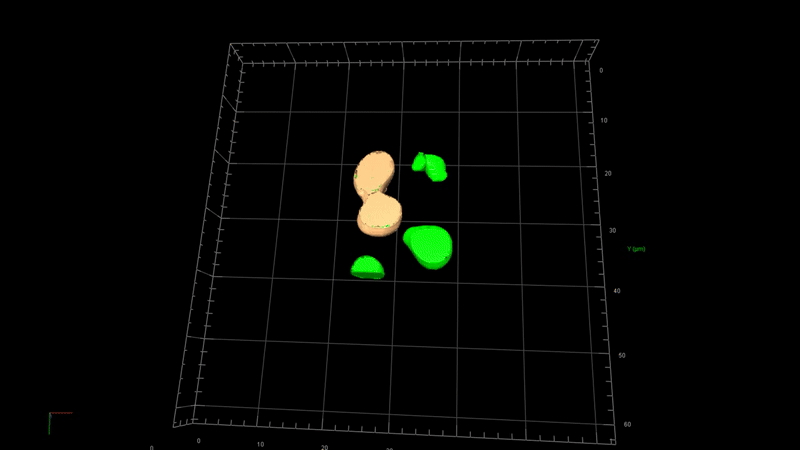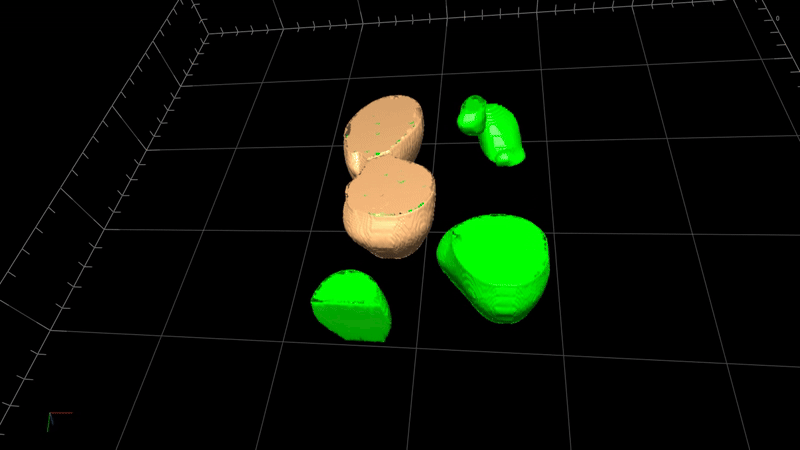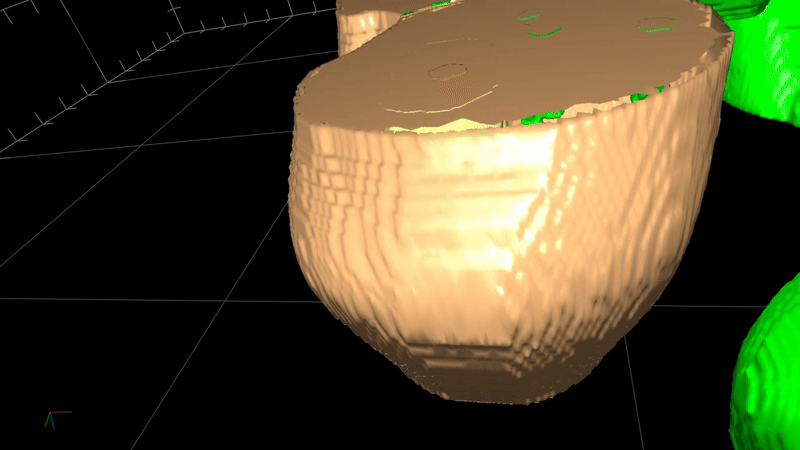
State-of-the-art image processing such as 2D/3D image analysis, 3D rendering, virtual reality
Image processing in microscopy is crucial because it enhances the ability to quantitively analyze, interpret, and extract meaningful information from microscopic images of biological samples. Advanced image processing software such as 2D/3D deconvolution offers several image processing techniques such as —
-
3D deconvolution
-
3D rendering
-
Virtual reality
-
Quantitative microscopy (intensity and volume measurements)
-
Image visualization and presentation
Image Courtesy: Dr. Gunjan Mehta
Specification of Microscope Available at Facility

Nikon Motorized Inverted Microscope Ti2E with Single-Molecule Localization Microscopy (SMLM/STORM) and SR-TIRF with Live Cell Imaging System
Specifications:
-
Objectives:
1) Plan Apochromat 20x with N.A 0.80
2) Plan Apochromat 40x with N.A 0.95
3) Plan Apochromat oil immersion 60x with N.A 1.42
4) Plan Apochromat/Apo 100x oil TIRF with N.A. 1.49 -
LED Light Source: Cool LED-pE-800 (365, 435, 470, 500, 550, 580, 635, 740 nm)
-
Lasers: 405 nm (fiber output power 50 mW), 488 nm (fiber output power 100 mW), 561 nm (fiber output power 70 mW), 640 nm (fiber output power 140 mW)
-
Epi-Fluorescence Filter cubes: highest quality bandpass fluorescent filter cubes for DAPI, CFP, YFP, GFP, TRITC, mCherry, CY5, and FRET (CFP-YFP).
-
Perfect Focus System (PFS)
-
Camera 1: ORCA-Fusion BT for STORM
-
Camera 2: Prime 95B
-
Motorized TIRF module for single-molecule imaging
-
Localization-based Super Resolution System for STORM/PALM/DNA PAINT
-
Software modules: NIS-ELEMENTS AR / CN, 3D Volume View 1, NIS-D Z DRIVE 1, NIS-A 3D Measurement 1, NIS-A 6D 1, NIS-A N-STORM Analysis 1, NIS-A RT Acquisition 1, NIS-A, Illumination Sequence 1, NIS-D Splitter DV 1, NIS-A CA FRET